|
The 1 s orbital at the bottom of the diagram is the orbital with electrons of lowest energy. Figure 6.24 depicts how these two trends in increasing energy relate. In any atom with two or more electrons, the repulsion between the electrons makes energies of subshells with different values of l differ so that the energy of the orbitals increases within a shell in the order s < p < d < f. The energy of atomic orbitals increases as the principal quantum number, n, increases. The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. This allows us to determine which orbitals are occupied by electrons in each atom. Having introduced the basics of atomic structure and quantum mechanics, we can use our understanding of quantum numbers to determine how atomic orbitals relate to one another. Relate electron configurations to element classifications in the periodic table.Identify and explain exceptions to predicted electron configurations for atoms and ions.Derive the predicted ground-state electron configurations of atoms.The number of protons determines an element’s identity.Īns.By the end of this section, you will be able to: The quantity of protons in an atom’s nucleus is known as the atomic number. It is a solid that has a lustrous grey colour and many of the same physical and chemical characteristics as the other five alkaline earth metals. The chemical element magnesium has the atomic number 12 and the letter Mg as its symbol. Barium is not a free element in nature due to its great chemical reactivity. It is a soft, silvery alkaline earth metal and the fifth element in group 2. Atomic Number of BariumĬhemical element barium has the chemical symbol Ba and atomic number 56. At room temperature, elemental sulphur is a crystalline solid that is brilliant yellow. Sulfur atoms normally combine to create cyclic octatomic molecules, which have the chemical formula S8. It is multivalent, nonmetallic, and plentiful. It belongs to the periodic table’s group 14 Atomic Number of SulphurĬhemical element sulphur has the letter S and atomic number 16. It is a tetravalent metalloid and semiconductor that has a blue-grey metallic sheen and is a hard, brittle crystalline solid. The crust of the Earth contains barely 0.025 percent carbon.Ītomic Mass Of Elements 1 To 30 With Symbol And PDF Atomic Number of SiliconĬhemical element silicon has the chemical symbol Si and atomic number 14. The periodic table’s group 14 includes it. It has a tetravalent atom, which means that four of its electrons can be used to create covalent chemical bonds. Atomic Number of CarbonĬhemical element carbon has the atomic number six and the letter C assigned to it. It makes up a large portion of both the Earth’s outer and inner core and is, by mass, the second-most prevalent element on Earth after oxygen. 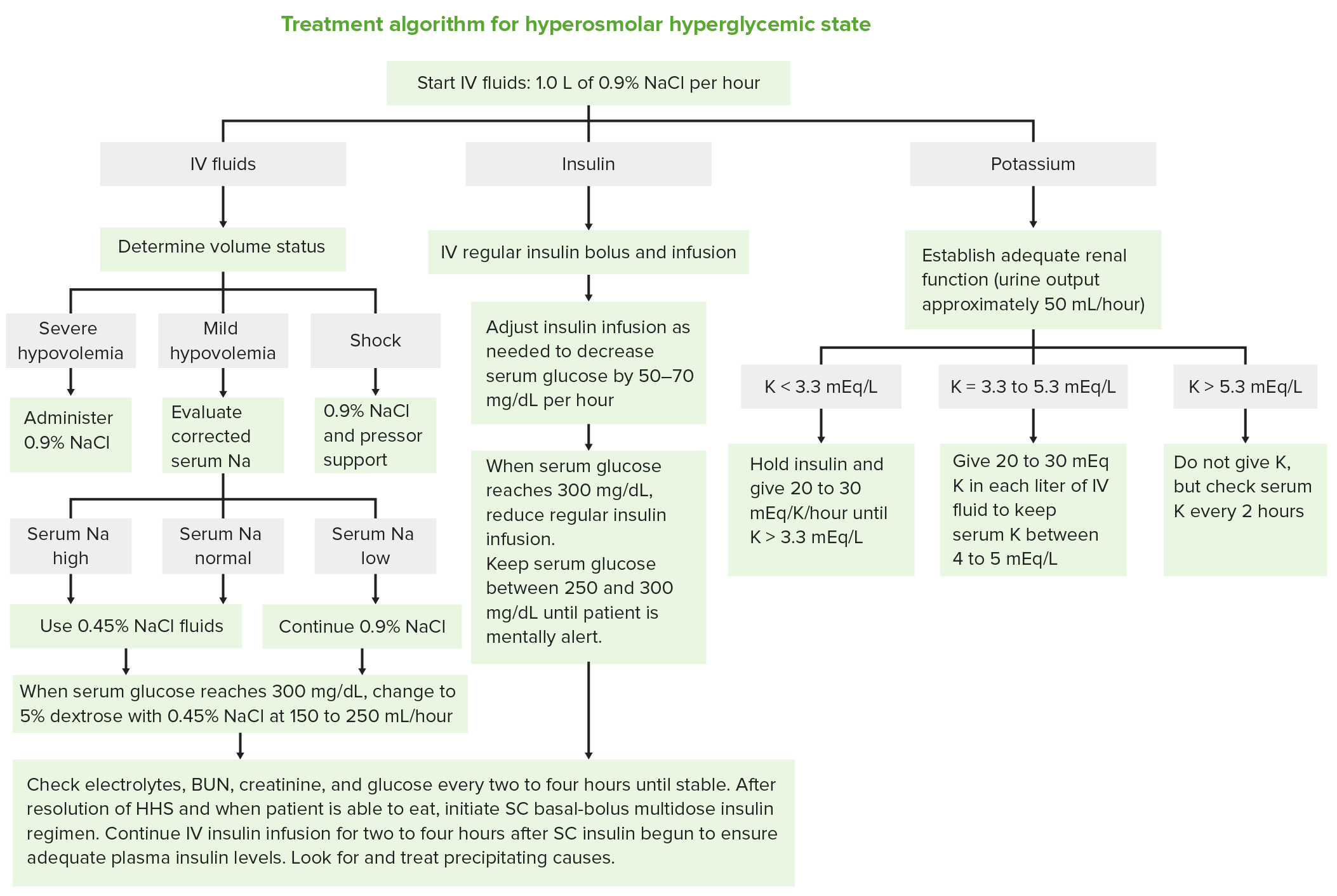
It is a metal that is found in group 8 of the periodic table and the first transition series. The chemical element iron has the atomic number 26 and the symbol Fe. It belongs to the periodic table’s chalcogen group, is a very reactive nonmetal, and an oxidising agent that easily produces oxides with most elements as well as other compounds. 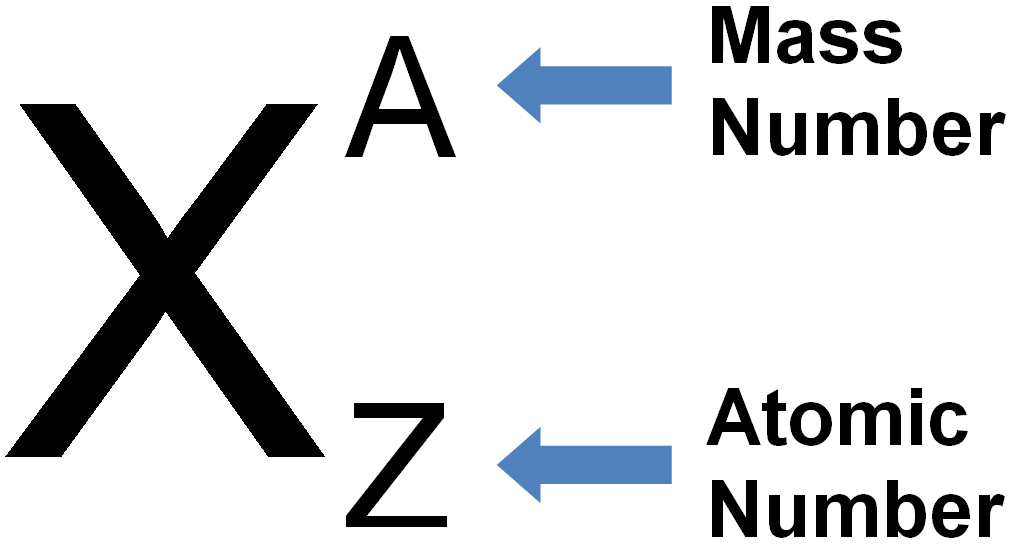 
The chemical element with the atomic number 8 and symbol O is called oxygen. It is a transition metal that is soft, white, and lustrous and has the highest electrical, thermal, and reflectivity of any metal.ĬaCO3- Chemical & Common Name, Molar & Atomic Mass Atomic Number of Oxygen The chemical element of silver has the atomic number 47 and the symbol Ag. Atomic Number Table and List Atomic Number The atomic number and the number of electrons are both equal in a regular, uncharged atom. Ordinary chemical elements can be uniquely identified by their atomic number. This is equivalent to the proton number (np), or the number of protons present in the nucleus of each atom of that element, for conventional nuclei. The charge number of an atomic nucleus is the chemical element’s atomic number, also known as nuclear charge number (symbol Z). NCERT Solutions Class 10 Social Science.NCERT Solutions For Statistics Class 11.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
